
With the rapid development of new prototypes and changes to how consumers view healthcare, the medical device market is growing fast. According to the latest market research, the industry will grow annually by 6.3% from 2021 to 2027, reaching a global market value of $625.3 billion. Below, we look at some of the medical device trends for 2022 driving this growth.
5 Medical Device Trends for 2022
New technologies and medical capabilities, including drug discovery and research are driving most of the growth..

by Lance Crawford, Thermal Press International, Inc.
Heat sealing is the process of sealing one thermoplastic to another similar thermoplastic using heat and pressure. The direct contact method of heat sealing utilizes a constantly heated die or sealing bar to apply heat to a specific contact area or path to seal or weld the thermoplastics together. Heat sealing is used for many applications, including heat seal connectors, thermally activated adhesives and film or foil sealing.
Common applications for the heat sealing process
Medical devices provide effective solutions for diagnosis, treatment, rehabilitation, and disease prevention. The size of the medical device market is expected to reach $623 billion by 2026 – a 5% compound annual growth rate. With help from the right plastic assembly process, your company can stay competitive in this growing market while also saving both time and money.
The most common plastic assembly processes used for medical devices include:
- Ultrasonic welding
- Vibration welding
Thermal Press International offers superior equipment and tooling for organizations that need reliable heat sealing and heat staking systems. We utilize high quality materials during production and take final products through a rigorous testing process to ensure they meet all product specification and production requirements.
The high ratings and reviews from clients prove the reliability and quality of our thermal plastic assembly machines. In a recent survey accomplished through telephone interviews, customers appreciated the positive impact of our machine on productivity, quality, and consistency.
So,..

Today, manufacturers in science and pharmaceuticals have an increased need for single-use bioreactor bags. Additionally, the recent pandemic has strained an already growing industry with the need for manufacturing vaccines around the world.
We have been a trusted source of thermal plastic assembly equipment here at Thermal Press International since 1976. As a result, we are a leader in the bioreactor bag marketplace, ready to meet the increased demands that today's manufacturing processes require.
Increased Demand for Bioreactor Bags
According to Precedence Research, medical device companies worldwide spent $53.71 billion on contract manufacturing in 2019 – and are expected to spend $97.52 billion annually by 2027. No doubt you're constantly examining your company's development costs, looking for ways to become more efficient.
Underestimating Assembly Costs
It's easy, especially in the planning stages, to underestimate manufacturing and assembly costs. It's natural to focus on the price of the components and assume that's going to be the bulk of your expenses...

While advances in plastic assembly techniques create innovative new products, the process also depends on the operators working efficiently at these stations. Establishing an effective development, test, and production process requires systems that conform to both commercial interests and human physiology.
Considerations for Deploying a Plastic Assembly Line in Your Company
Often overlooked during the product development and design stages, the configuration of plastic assembly stations can have a major impact on your company’s productivity. The long-term effects of working..
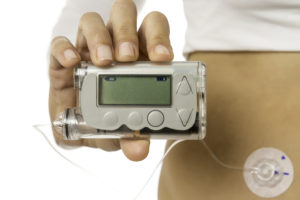
Modern medicine has come a long way, and today we have a variety of applications in the medical industry that depend on plastic assemblies. The increasing role that technology plays in providing quality healthcare and automated medicine delivery to patients requires a high-quality plastic assembly process.
Wearable medical devices with integrated displays help physicians monitor and log a patient’s health conditions, sometimes integrating directly with their electronic health records (EHR). In an evolving market, more practitioners now depend on sophisticated electronic products to deliver precise doses of medicine. The

At Thermal Press, we understand that a quality assembly process is vital for today’s consumer and medical product industries. When it comes to a new product introduction (NPI) or prototyping project, you’ll need to work with a team of experts that have years of industry experience and access to the latest technologies.
Thermal Press is a leader in plastic assembly and heat sealing solutions. With a range of platforms and configurations available, we will work with your product team for their plastic assembly process development requirements. We can customize our..

Advances in technology have opened new vistas for medical devices and fluid delivery systems. These solutions integrate with human biology to deliver medicine or extract fluids safely and hygienically. Heat sealing plastics made these innovations possible by providing hermetically sealed plastic delivery systems that remain safe to the wearer.
In situations where ultrasonic welding isn’t suitable, heat sealing provides a method to overcome challenges by creating a secure bond between thermoplastics using precise heat and pressure. Although the applications for these types of seals vary, the market continues to grow..






